 
VSEPR theory also states that the lone pair of electrons repels bonding pairs more than bonding-bonding pair repulsion, and so you can imagine the 1 lone pair squeezing the bonding pairs slightly closer together in NH3, which reduces the bond angle by about 2 degrees, making the bond angle 107.5 degrees. NH3 is made from three N-H bonds with one lone pair on the nitrogen atom. However, as only 3 of the electron pairs are bonding, there isn't a bond at the 'top' to form a tetrahedron, and the actual shape of NH3 is trigonal pyrimidal. The NH3 molecule has a trigonal pyramidal form, with a bond angle of 107.3 degrees. As there are 4 electron pairs around the central nitrogen, you can imagine each spreading out to the 4 corners of a tetrahedron, which would suggest that the bond angle is 109.5 degrees. The valence shell electron pair repulsion (VSEPR) theory states that electron pairs around a central atom will repel so they are as far away as possible. 5-3=2, so that leaves 1 lone pair of electrons around this centrral nitrogen atom. As covalent bonds involve the sharing of electrons, and there are 3 covalent bonds, nitrogen shares 3 of its 5 outer electrons, 1 each with the 3 hydrogens, and so there are 3 bonding pairs. As hydrogen can only ever have 1 bond to/from it, we can say that nitrogen is the central atom with hydrogens around it. B.So if we think about a molecule of ammonia, NH3, there are 3 hydrogens, each with 1 electron (as its atomic number is 1) covalently bonded to 1 nitrogen atom with 5 outer shell electrons on it due to its atomic number of 7, but having 2 electrons in its innermost shell. Medvedev, V.A.CODATA Key Values for Thermodynamics. 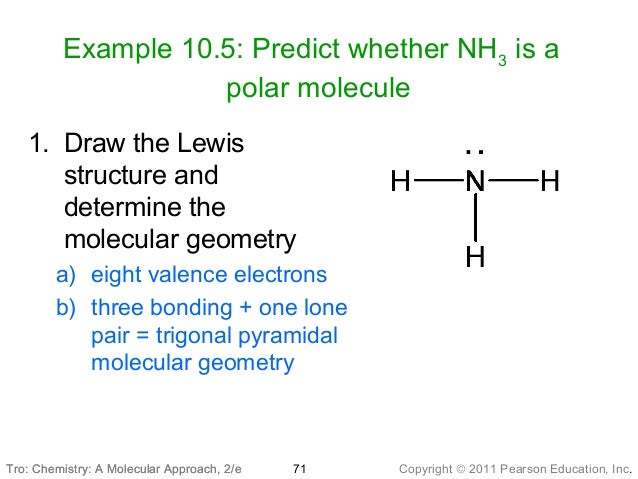 
TN Olney, NM Cann, G Cooper, CE Brion, Absolute scale determination for photoabsorption spectra and the calculation of molecular properties using dipole sum-rules, Chem. Th Koops, T Visser, WMA Smit "Measurement and Interpretation of the Absolute Infrared Intensities of NH3 and ND3" Journal of Molecular Structure 96 (1983) 203-218 Landolt-Bornstein: Group II: Volume 6 Molecular Constants from Microwave, Molecular Beam, and Electron Spin Resonance Spectroscopy Springer-Verlag. Please address comments about this page to G., Electronic spectra and electronic structure of polyatomic molecules,Van Nostrand,New York, 1966į Shimizu "Stark Spectroscopy of NH3 ν2 Band by 10-μ CO2 and N2O Lasers" J. NIST does not necessarily endorse the views expressed, or concur with the facts presented on these sites.įurther, NIST does not endorse any commercial products that may be mentioned on these sites. There may be other web sites that are more appropriate for your purpose. No inferences should be drawn on account of other sites being referenced, or not, from this page. We have provided these links to other web sites because they may have information that would be of interest to you. Vibration specific dipole moments Vibrational Quantum numbersĬalculated electric quadrupole moments forĮlectric dipole polarizability (Å 3) alphaĬalculated electric dipole polarizability forīy selecting the following links, you may be leaving NIST webspace. You are here: Experimental > One molecule all propertiesĪmmonia Ammonia gas Ammonia, anhydrous Anhydrous ammonia Aromatic Ammonia, Vaporole Nitro-Sil Spirit of Hartshorn azane Įxperimental dipole measurement abbreviations: MW microwave DT Dielectric with Temperature variation DR Indirect (usually an upper limit) MB Molecular beam 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
